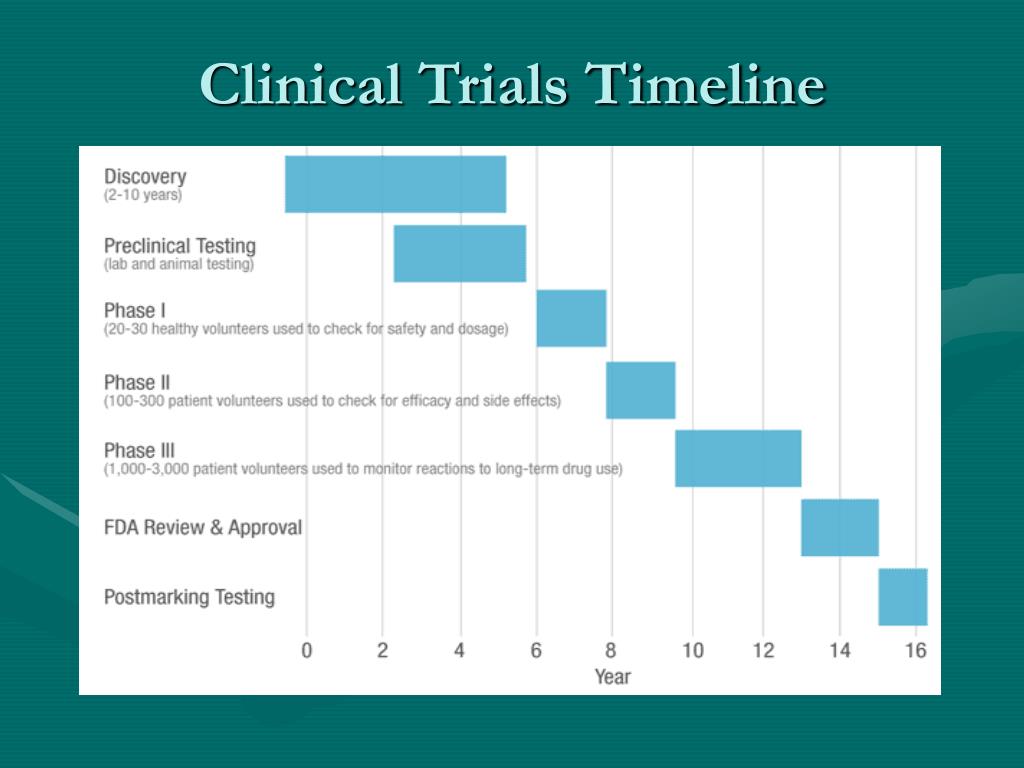
Only 32% of respondents were found to have consistent cycle times across all study start-up activities, completing site activation about 7 weeks earlier than other companies when working with both new and repeat investigative sites. So, what attributes make some organizations more consistent that others in adherence to timelines and budgets? Planning works by getting it right from the beginning - prior to study activation - requiring sponsors and CROs to identify what is needed upfront to reduce risk. These words ring true when it comes to study start-up, especially as the clinical trials sector embraces planning as key to boosting study quality. If you fail to plan, you are planning to fail. Use of site-level data to predict enrollment may be a more attractive option for increasing the pool of evidence available to support study start-up decision-making. Although clinical research professionals recently have been seeking access to accurate site-level performance metrics to aid investigative site identification. 4Īccording to the research, companies do not use one single source of data to identify sites a mix of non-evidence-based approaches are used, including personal networks, proprietary databases, and recommendations from study teams. Moreover, study teams are blinded to problems inherent with this approach –namely, it limits opportunities to engage with new sites that could be more effective than those familiar to the study team. Institutional knowledge about sites is frequently dated and soiled within departments and may not be relevant to the therapeutic area under investigation, for example, rare and orphan disease trials often require companies to work with sites and investigators they have not interacted with in the past. Clinical operations teams typically rely on relationships with principal investigators built over time, and while the idea of using all repeat sites might seem like a logical and sure-win way to speed study start-up it is important to point out that research suggests for a typical multi- center study, 30% of sites selected are new, of which 13% are completely new to clinical research. The research found that cycle times were shorter for repeat sites than they were for new sites (Figure 1). Study start-up (also referred to as site readiness and site activation) cycle time was measured as the time that all initial sites (ie, non-backup or contingency sites) are activated or from the time the site selection decision is made until all sites are initiated and ready to enroll. Site selection cycle time was defined as the time from site identification to feasibility and receipt of site qualification information to final site selection decision. Site identification cycle time was defined as the time taken to identify appropriate investigative sites. 
More than half of respondents worked in sponsor companies 53% or CROs 24%, with additional responses from sites, medical device companies, and academic institutions. 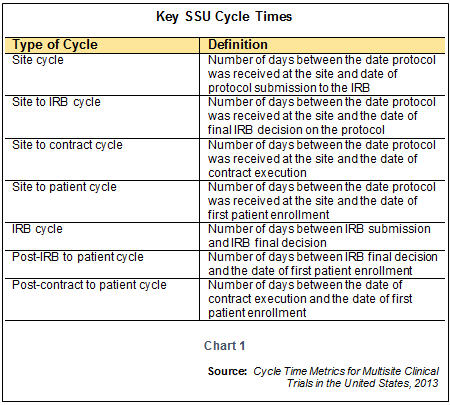
The respondents were composed of 403 unique organizations and three-quarters were US-based. The research examined a number of areas associated with study start-up, including site identification, study feasibility and recruitment planning, criteria for site selection, staffing and resources, and study start-up process improvements and opportunities. The study was funded by an unrestricted grant from goBalto, a technology solutions provider. Given the plethora of new approaches and solutions now being deployed to improve the study start-up process, the Tufts CSDD research provides a baseline upon which future studies can be conducted to gauge progress. 3 Given the new technology solutions and practices, as well as the increasing number of dedicated personnel managing site relationships, it’s surprising and disappointing that the industry has not been about to make any headway in reducing the number of non-active, non-enrolling (NANE) sites. 1 The primary reason cited was budgeting and contracting problems, which has been a challenge identified in much published work. In a recently completed comprehensive study, The Start-Up Time and Readiness Tracking (START) II, 2017 conducted by Tufts Center for the Study of Drug Development (CSDD), a significant difference in cycle times between new versus repeat sites and organizations was observed however, the percentage of sites never activated remained at 11%, a figure that has not changed substantially in over a decade. As stakeholders are increasingly aware that better study startup (encompassing the activities associated with site identification, feasibility assessment, selection, and activation) processes are linked to shorter clinical timelines, the emphasis has been shifting in that direction.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
